
Another way of stating this is that the structure consists of two interpenetrating fcc lattices, which are related to each other by a translation of half the unit cell along any of the three Cartesian axes. Note that each of the Na + positions is related to a Cl - position by a translation of (1/2 0 0). There are four of these holes per cell, and they are filled by the chloride ions. The octahedral holes in a face-centered cubic lattice can be found at fractional coordinates (1/2 1/2 1/2), (1/2 0 0), (0 1/2 0), and (0 0 1/2). In the NaCl structure, shown on the right, the green spheres are the Cl - ions and the gray spheres are the Na + ions. Each cell contains four packing atoms (gray), four octahedral sites (pink), and eight tetrahedral sites (blue). One octahedral and one tetrahedral site in a face-centered cubic unit cell. 
The NaCl structure is a good example of the latter. While this is often true, there are many examples of structures in which cations are the packing atoms, and others in which the distinction is arbitrary. We might expect that anions, which are often larger than cations, would be better suited to the positions of packing atoms. Question: Would anions or cations be better as packing atoms? In both the hexagonal close packed and cubic close packed lattices, there is one octahedral hole and two tetrahedral holes per packing atom. An interstitial atom filling a tetrahedral hole is coordinated to four packing atoms, and an atom filling an octahedral hole is coordinated to six packing atoms. These packing lattices contain two types of sites or "holes" that the interstitial atoms fill, and the coordination geometry of these sites is either tetrahedral or octahedral. 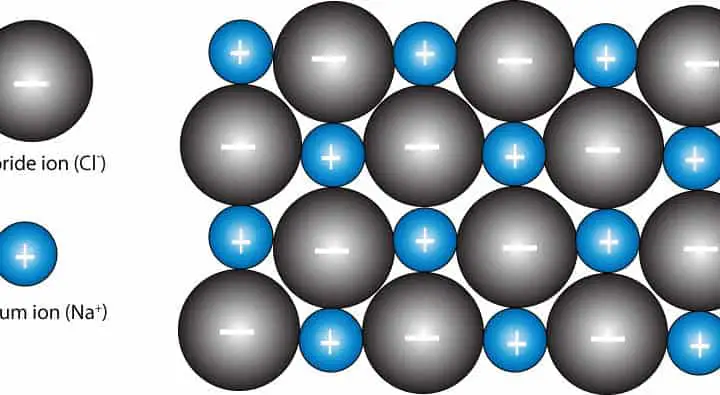
Many common inorganic crystals have structures that are related to cubic close packed (face-centered cubic) or hexagonal close packed sphere packings.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
